State the basic postulates of Bohr atomic model and derive an expression for wave number of radiations emitted by an - Physics - Atoms - 14247985 | Meritnation.com

Bohr's atomic model for class 11 (Inter) in telugu || Bohr's postulates || - YouTube | Class, Atom, Telugu

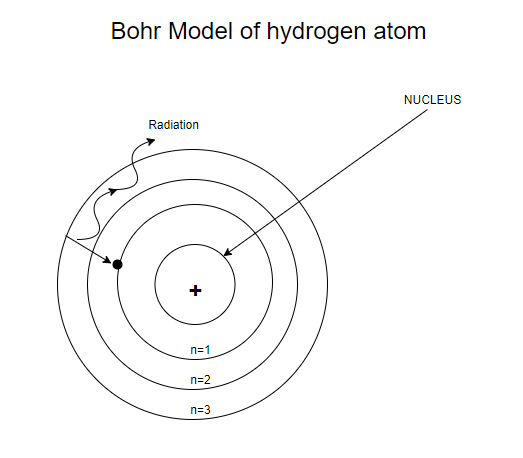
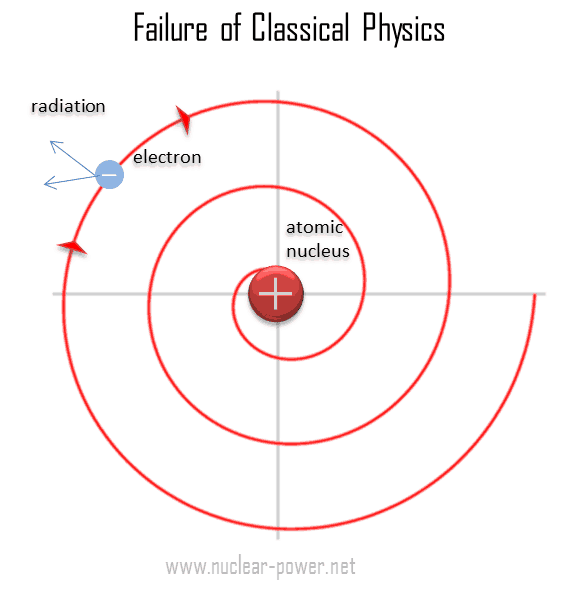
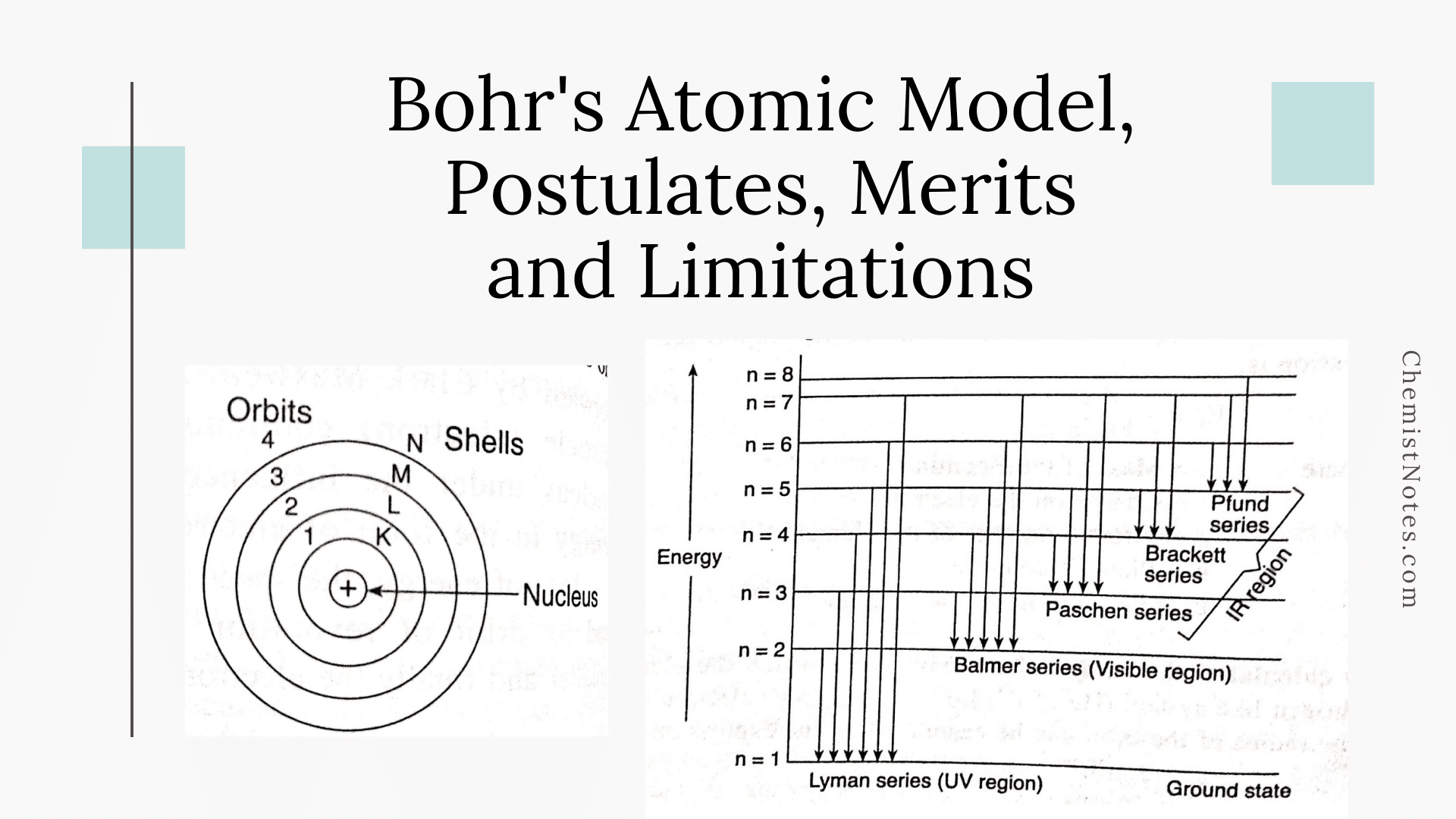
State Bohr's postulates. Using these postulates, derive an expression for total energy of an electron in the n^("th") orbit of an atom. What does negative of this energy signify ?